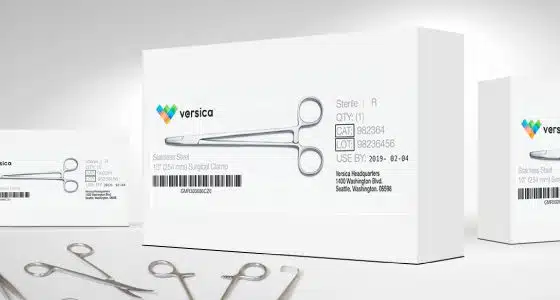
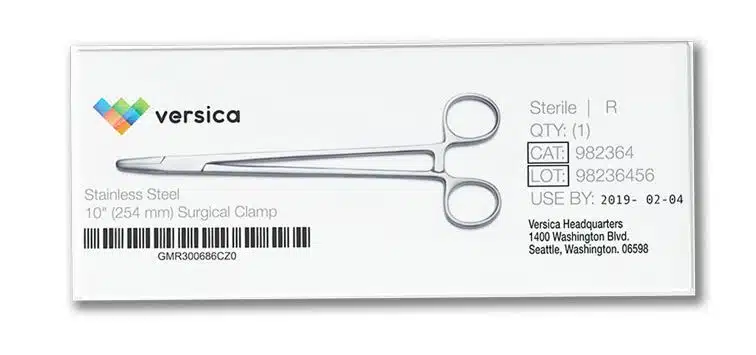
 AstroNova has a long history of working closely with the medical device and medical supply community for over 30 years, including experience in achieving ISO 13485 certification and producing a variety of medical devices and supplies of our own.
AstroNova has a long history of working closely with the medical device and medical supply community for over 30 years, including experience in achieving ISO 13485 certification and producing a variety of medical devices and supplies of our own.
Due to recent events, we are receiving requests for expedited orders to help our customers who are actively involved in the fight against COVID-19. One customer specifically, a major American manufacturer of diagnostic healthcare products, is working diligently to produce swab collection kits, among other products to provide to labs and the government. To meet this demand, they are exponentially ramping up production to meet the needs. In short, every piece produced needs a label.
How are AstroNova Printers and Materials Qualified for the Medical Community?
Over the years AstroNova has worked with leading medical device manufacturers to ensure AstroNova label printers and label materials pass mandated, stringent regulatory requirements and independent laboratory testing to support their global packaging initiatives on these key points:
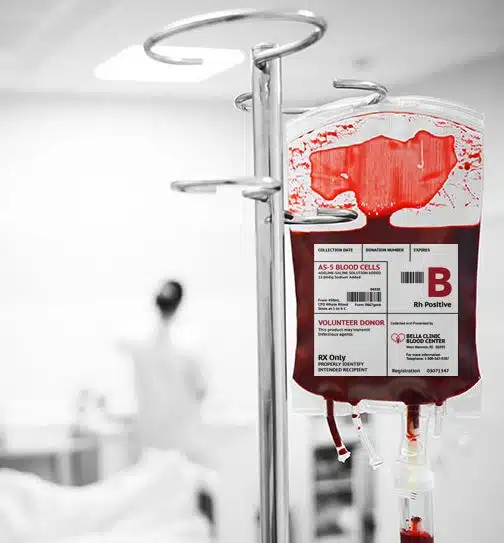 Establish AstroNova printers that can effectively, accurately, and repeatedly produce labels with adequate color matching
Establish AstroNova printers that can effectively, accurately, and repeatedly produce labels with adequate color matching- Substantiate that the inkjet ink, thermal transfer ribbons, and/or toners used by AstroNova label printers provide adequate abrasion resistance, moisture & chemical resistance, ensuring high-quality readability, free of excessive scuffing that might prevent legibility or barcode scanning.
- To show label stock sufficiently adheres to a variety of packing materials and substrates used in many packaging configurations including pouches, trays, bags and tubes, both sterile and non-sterile materials such as foil, Tyvek® (some corrugate and some with SBS – sterile barrier system), film, bubble bag, polybag, and tube and furthermore, remained adhered to their package component
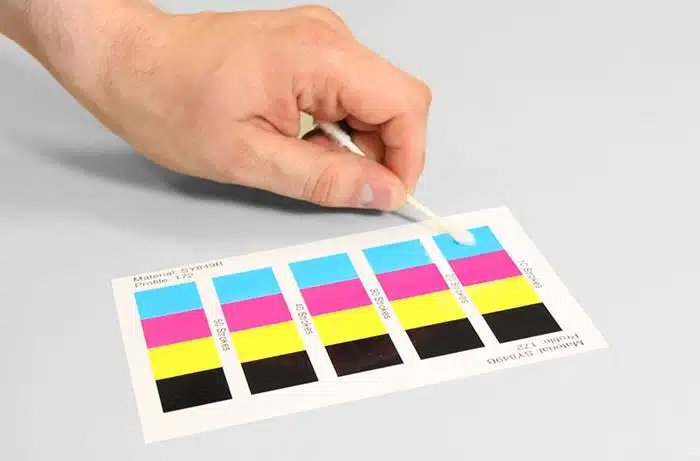 In conclusion, our samples passed both the initial and post-conditioning review with a 100% pass rate with 0% failures, validating AstroNova label printers and AstroNova label stock successfully meeting the required protocol and passing all validation requirements. Thereby, the validation team was able to confirm that the AstroNova label printer, and AstroNova label stock combination is a proven system to support the sterile and non-sterile box and label operations and the Global Label initiative.
In conclusion, our samples passed both the initial and post-conditioning review with a 100% pass rate with 0% failures, validating AstroNova label printers and AstroNova label stock successfully meeting the required protocol and passing all validation requirements. Thereby, the validation team was able to confirm that the AstroNova label printer, and AstroNova label stock combination is a proven system to support the sterile and non-sterile box and label operations and the Global Label initiative.
How Does it Differ from Verification?
Process validation is a term used in the Pharmaceutical and Medical Device industries to indicate that a process has been subject to such scrutiny that the result of the process (a product, service, or other outcomes) can be practically guaranteed. The general public may use terms like “verification” or “validation” loosely or interchangeably in conversations, but the FDA assigns specific definitions to terms like these.
For example:
Validation means confirmation by examination and provision of objective evidence that the requirements for specific intended use can be consistently fulfilled. [Definitions 21 CFR 820.3 (z)]
Verification means confirmation by examination and provision of objective evidence that specified requirements have been fulfilled. [Definitions 21 CFR 820.3 (aa)]
While the completion of process validation is a regulatory requirement, a manufacturer may decide to validate a process to improve overall quality, eliminate scrap, reduce costs, improve customer satisfaction, or several other reasons. Coupled with adequately controlled design and development activities, a validated process may result in a reduced time to market for new products. The Pharmaceutical and Medical Device industries are facing an unprecedented situation right now due to the COVID-19 pandemic, which has impacted communities all across the world. Time to market, while always deemed necessary by the industry, is even more critical now as millions of lives can be potentially impacted.
Phases of Process Validation
In general, the validation of a process is the mechanism or system used by the manufacturer to plan, obtain, record, and interpret data. These activities may be considered to fall into three phases:
- An initial qualification of the equipment used and provision of necessary services – also known as installation qualification (IQ)
- A demonstration that the process will produce acceptable results and establishment of limits of the process parameters – also known as an operational qualification (OQ)
- Establishment of long-term process stability – also known as performance qualification (PQ).
Since our label printer solutions are used widely in the Pharmaceutical and Medical Device industries, AstroNova understands these challenges and can support our customers with these process validations on our printer products and supplies, ensuring they can be completed smoothly and quickly.
We just released procedures and templates to help perform IQ, OQ, and PQ on our QL-120/120X range of printers. Our trained technical support staff can help your teams complete these procedures remotely as well as ensure that process validations can be done in a safe manner, and quality products can be produced and shipped out of your facilities in record time.
For more information about Process Validations and IQ, OQ, PQ, please visit:




